TECHNICAL INFORMATION
Provides technical information related to products by AQUOSYS.
HOME
TECHNICAL INFORMATION
PFAS Treatment
PFAS Treatment
- > Overview
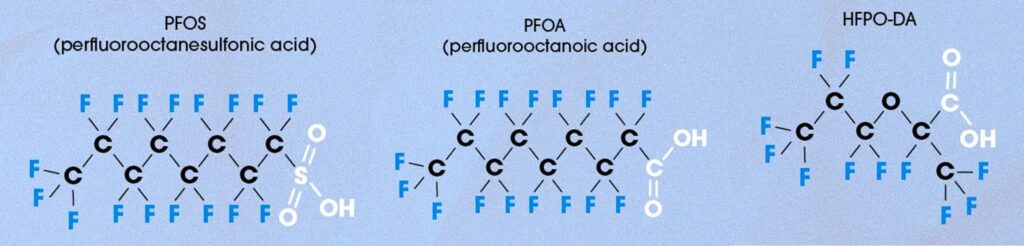
- PFAS are organofluorine compounds that, unlike typical organic compounds, have carbon-fluorine bonds instead of carbon-hydrogen bonds within their molecules.
These compounds are chemically very stable and difficult to decompose due to the strong bond between carbon and fluorine within the molecule.
PFAS possess low affinity for both water and oil, are heat-resistant, and are resistant to chemical attack. They have been widely used worldwide for a long time in a wide range of applications, including anti-adhesives, polishes, paints, and chemical-resistant materials.
However, with the recent discovery of PFAS’s ecological accumulation and toxicity, humanity has become aware of the serious threat and begun developing methods to remove them from the natural environment.
- ㆍ Chemical structures of some PFAS
- ㆍ PFAS Regulatory Standards by Major Countries (Drinking Water)
- > Treatment Technologies PFAS are extremely poorly biodegradable, making them difficult to break down naturally in the natural environment or remove through biological treatment processes. Therefore, physicochemical treatment technologies are employed.
- > AQUOSYS’s PFAS Treatment Technology AQUOSYS’s PFAS treatment technology utilizes the principle of foam separation using HELIFLOT. This involves mixing fine-bubbles with the raw water to be treated, then creating a swirling upward flow within a cylindrical foam separation tank. This causes the PFAS components adsorbed on the fine-bubbles to form and float to the water surface, where they are then removed.
Legislated PFAS water quality standards
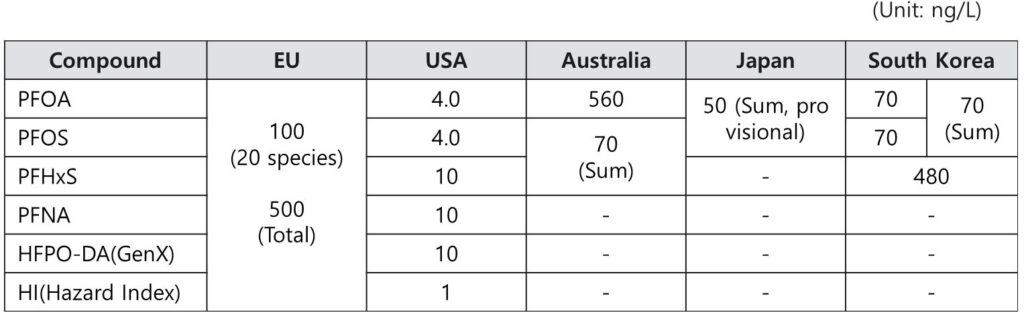
※ US: Applicable from 2031, EU/Japan: Applicable from 2026
ㆍ Activated Carbon Adsorption : Activated carbon, a material with numerous pores within its interior, is known for its outstanding ability to adsorb and remove various hazardous substances and odor molecules in liquids. Numerous experimental studies have shown that activated carbon is also effective in removing PFAS, and the U.S. Environmental Protection Agency (EPA) recommends activated carbon adsorption as a treatment technology for PFAS in drinking water.
However, a practical challenge for its application is the high cost of treating tap water using activated carbon.
ㆍ Ion Exchange : Most PFAS compounds have a chemical structure of carboxylic acid or sulfonic acid. Therefore, these compounds can be treated using ion exchange. This method is also recommended by the EPA as a treatment technology for PFAS in drinking water. However, this method is more expensive than the activated carbon adsorption method mentioned above.
ㆍ Membrane Filtration : This method removes PFAS compounds using a fine filtration membrane, such as a reverse osmosis membrane. This method is also recommended by the EPA as a treatment technology for PFAS in drinking water. However, this method is more expensive than the ion exchange method mentioned above.
ㆍ Foam fractionation : Most PFAS substances contain both hydrophilic and hydrophobic groups within their molecules. Regulated substances, in particular, possess hydrophobic groups with 6 to 9 carbon atoms attached to carboxylic or sulfonic acid groups. This allows them to function as surfactants. Indeed, they are often used as emulsifiers or foaming agents. Therefore, these substances can be treated using foam-based adsorption and fractionation. However, most companies offering foam fractionation technologies propose injecting an additional hydrocarbon surfactant into the treatment process.
While this method is relatively economical, it has limitations in reducing the final residual PFAS concentration below environmental regulatory limits, often requiring the addition of activated carbon adsorption stage at the end of the treatment process.
ㆍ Other methods : Oxidation treatment methods using ozone or electric electrodes are being considered for directly treating PFAS in water. However, as mentioned earlier, PFAS are fundamentally chemically stable substances, so their reaction rate with oxidizing agents is very slow, making efficient treatment difficult.
This method enables the removal of PFAS without the use of auxiliary surfactants or other chemicals.
This system not only realizes large-scale facilities, but is also highly cost-effective because it uses only air bubbles.